3D tissue models: Upscaling in vitro tests
Electric Impedance Sensing as a Readout of Organoid Viability

This collaborative project with Nanion Technologies and the Max Reichert group at Klinikum rechts der Isar is directed towards the investigation of Printed Circuit Board (PCB) technologies for their suitability to achieve a sensor-based readout of 3D cell models such as spheroids or organoids. There is a widespread use of both kinds of 3D cell models for preclinical and clinical research purposes. Fig. 1 shows a microscope image of an organoid culture.
One of the most beneficial features of a sensor-based, kinetic data readout is the ability to monitor the condition of 3D cell models individually and throughout the entire growth and drug incubation period. In contrast to endpoint measurements this allows additional longitudinal observations.
Multilayer PCB technology offers high-density integration of electrode structures for electric impedance recordings, industrial upscaling with low product costs and biocompatible assemblies (Fig. 2).
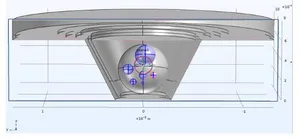
The planning of PCB design usually starts with virtual prototyping, where different kinds of electrode setups and geometries are tested as finite element models (Fig. 3).

One of the challenges of measuring the electric impedance of 3D culture objects is to guide electric currents through the 3D objects of interest and to avoid bypassing of high-impedance volumes. One of the approaches to achieve that is the use of guardring electrodes equipotential to the measuring working electrodes. Due to an altered electric field distribution with active guardring electrode, an increased share of working electrode current lines (in red) passes through the 3D object of interest. Depending on the geometry of measurement, the sensitivity of detecting biological changes will be enhanced (Fig. 4).
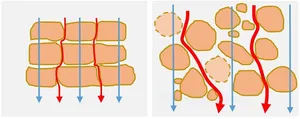
Electric impedance measured in-vitro on 3D cellular structures in the 10 kHz frequency range will respond to changes of bulk electrical conductivity (e.g. due to cell metabolism) and structural changes that can be related to alterations or damages of cell-cell connections and cell membrane integrity (Fig. 5).
Contact: Dr. Martin Brischwein, brischwein@tum.de
